DISSOLVING PROCESS
If we want to understand solutions, we must first examine how solutions are formed. In this lesson, we will learn about the dissolving process; more specifically the dissolving of a solid in a liquid.
Watch this video and make notes on the following key ideas:
1. What is electronegativity?
2. compare polar and non-polar molecules.
3. Explain and give examples of covalent bonds, polar covalent bonds, and ionic bonds.
4. Why do Non-Polar molecules not mix with water?
5. How does soap work?
1. What is electronegativity?
2. compare polar and non-polar molecules.
3. Explain and give examples of covalent bonds, polar covalent bonds, and ionic bonds.
4. Why do Non-Polar molecules not mix with water?
5. How does soap work?
The Dissolving Process
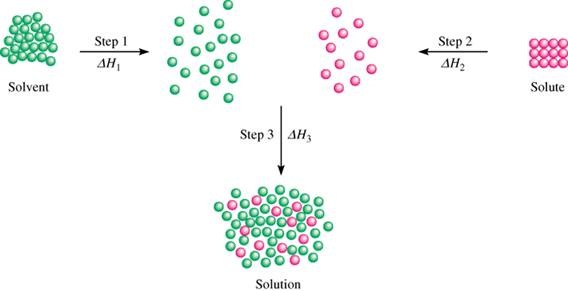
When a solute dissolves in a solvent the individual particles of the solute separate from the other particles of the solute and move between the spaces of the solvent particles. The solvent particles must collide with the solute particles and forces of attraction between solute and solvent particles "hold" the solute particles in the spaces. See the diagram below.
There are 3 steps to the dissolving process:
1. The solvent particles must move apart to make room for solute particles.
This process requires energy to overcome forces of attraction between solvent particles.
The first step in the dissolving process is endothermic.
2. The solute particles must separate form the other solute particles.
This process also requires energy to overcome the forces of attraction between the solute particles.
The second step in the dissolving process is endothermic.
The energy of this step is known as lattice energy.
Lattice energy is the amount of energy required to separate the molecules or ions from each other in
a solid crystal.
3. When the solute particles move between the solvent particles the forces of attraction between solute and solvent take hold and the particles "snap" back and move closer.
This process releases energy.
The final step in the dissolving process is exothermic.
This is known as the heat or energy of hydration.
When water surrounds individual molecules or ions and the molecules or ions are said to be hydrated.
When a solvent other than water is used the processes of the solvent particles surrounding the
solute particles is called solvation.
Energy Changes During Dissolving
The total heat change in the dissolving process is called the heat of solution.
The heat of solution is equal to the sum of the heat changes for the three steps in the dissolving process.
If the sum of the heat absorbed in the first two steps of the dissolving process is greater than the heat released in the last step, the dissolving of that substance will be endothermic.
If the dissolving process for a substance is endothermic, the container will feel cooler as the substance dissolves.
The dissolving of most solids in water is an endothermic process.
The endothermic dissolving process is used in cold packs, often found in first aid kits. The cold pack is a bag containing a solid or a liquid with a smaller bag or vial in the larger bag. The cold pack is activated by breaking open the inner vial. Breaking open the vial releases allows solid ammonium nitrate to mix with the liquid, usually water. The
dissolving of ammonium nitrate absorbs large amounts of heat form its surroundings including the skin it is placed against.
In some cases, the exothermic process is larger than the sum of the two endothermic processes.
In these cases, as the solid dissolves large amounts of heat are evolved.
Examples of these types of solids are calcium chloride, sodium hydroxide and potassium hydroxide.
Hot packs found in first aid kits will often contain a larger bag of solid calcium chloride with a smaller inner bag of water. Breaking the inner bag releases water into the calcium chloride. As the calcium chloride dissolves, it
releases heat.
The total heat change in the dissolving process is called the heat of solution.
The heat of solution is equal to the sum of the heat changes for the three steps in the dissolving process.
If the sum of the heat absorbed in the first two steps of the dissolving process is greater than the heat released in the last step, the dissolving of that substance will be endothermic.
If the dissolving process for a substance is endothermic, the container will feel cooler as the substance dissolves.
The dissolving of most solids in water is an endothermic process.
The endothermic dissolving process is used in cold packs, often found in first aid kits. The cold pack is a bag containing a solid or a liquid with a smaller bag or vial in the larger bag. The cold pack is activated by breaking open the inner vial. Breaking open the vial releases allows solid ammonium nitrate to mix with the liquid, usually water. The
dissolving of ammonium nitrate absorbs large amounts of heat form its surroundings including the skin it is placed against.
In some cases, the exothermic process is larger than the sum of the two endothermic processes.
In these cases, as the solid dissolves large amounts of heat are evolved.
Examples of these types of solids are calcium chloride, sodium hydroxide and potassium hydroxide.
Hot packs found in first aid kits will often contain a larger bag of solid calcium chloride with a smaller inner bag of water. Breaking the inner bag releases water into the calcium chloride. As the calcium chloride dissolves, it
releases heat.
Watch this video and make notes on how dissolving works. Make sure to differentiate how Ionic compounds dissolve and how Covalent compounds dissolve.
Dissociation Equations
Example 1. Write the equation for dissolving solid magnesium chloride, MgCl2, in water.
Example 2. Write the equation for solid aluminum sulphate, Al2(SO4)3 dissolved in water.
Example 3. Write the equation for the dissolving of liquid methanol, CH3OH, in water.
Example 1. Write the equation for dissolving solid magnesium chloride, MgCl2, in water.
Example 2. Write the equation for solid aluminum sulphate, Al2(SO4)3 dissolved in water.
Example 3. Write the equation for the dissolving of liquid methanol, CH3OH, in water.